WELCOME TO DEEP-TECH INDUSTRIES 4.0
We Provide One Platform – Multi Layers of Intelligence
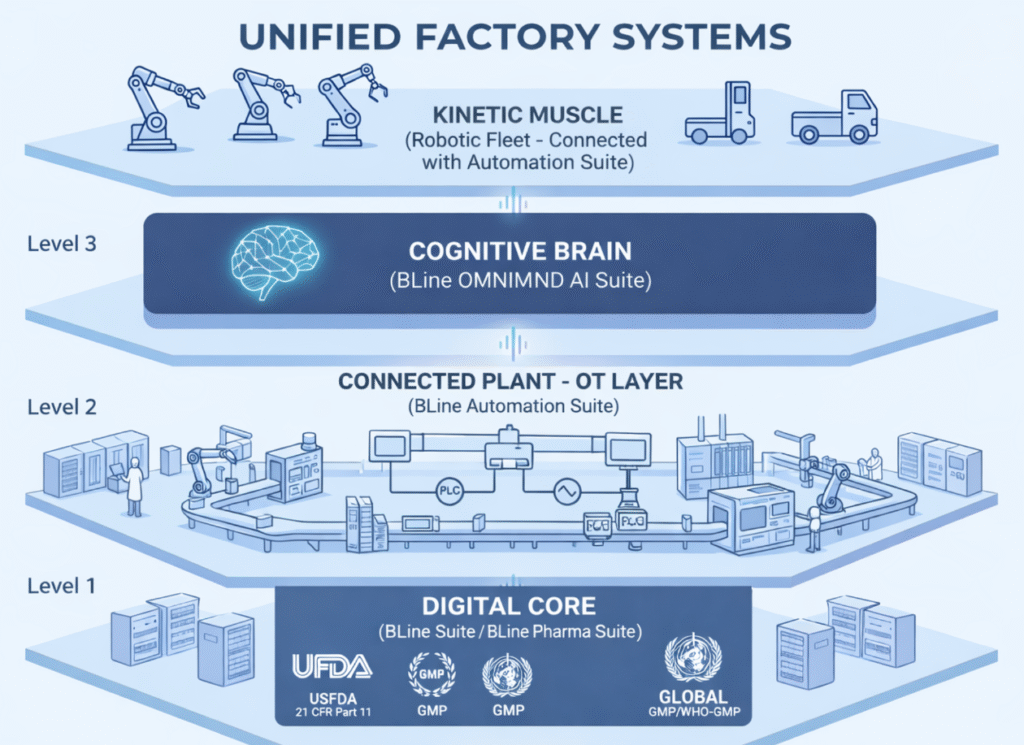
Level 1: The Digital Core (BLine Suite/BLine Pharma Suite)
The foundation of paperless operations. We ensure data is “Born Digital” and remains audit-ready in real-time, whether you are tracking life-saving drugs or high-precision components.
Level 2: The Connected Plant – OT Layer (BLine Automation Suite)
Bridging the IT-OT gap. We synchronize enterprise data with plant machinery via PLC/SCADA integration, ensuring real-time data exchange and Industry 4.0 readiness.
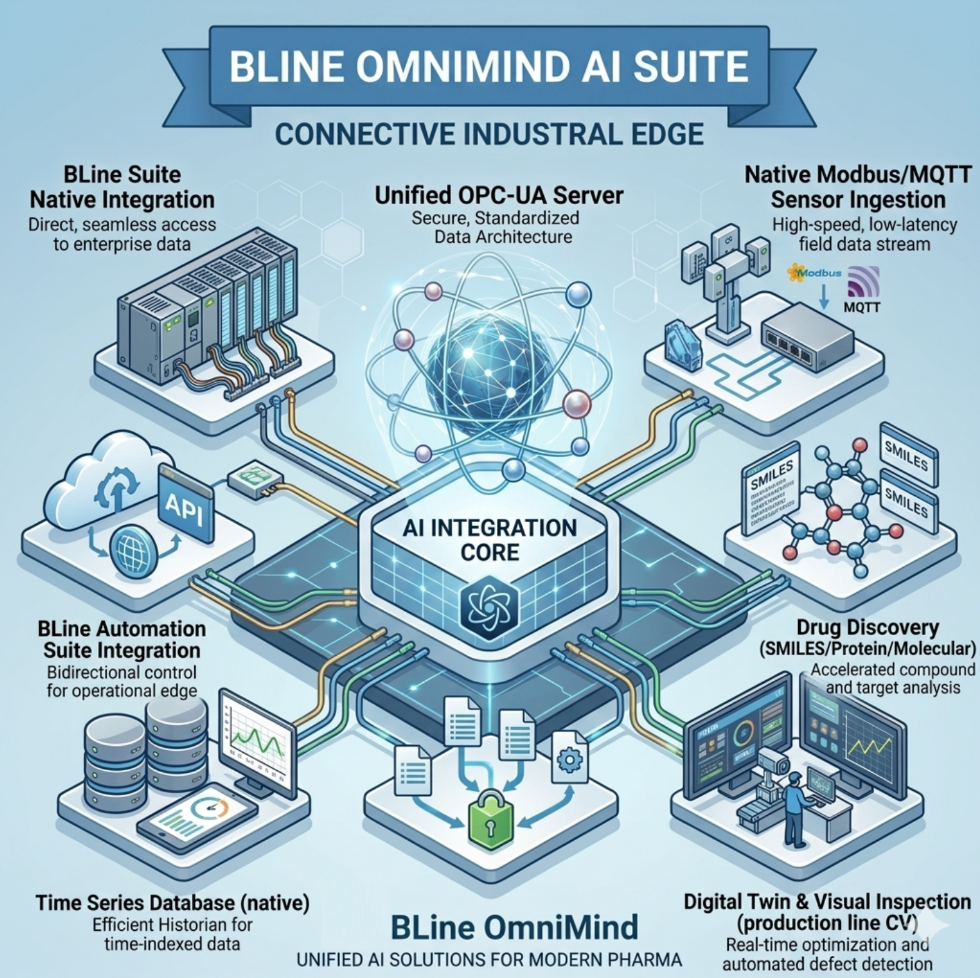
Level 3: The Cognitive Brain (BLINE OMNIMIND AI Suite)
Turning data into foresight. Our AI monitoring, smart vision cameras, and predictive analytics optimize process quality, security, and activity tracking across any production line.
Level 4: The Kinetic Muscle (Robotic Fleet – Connected with Automtion Suite)
The final frontier of automation. We deploy AGVs and AMRs for autonomous material handling, eliminating human error and contamination risks in sensitive environments.
AI BASED SOLUTION – From Data to Delivery Fully Automated
BLine Solution transforms manufacturing plants and factories into fully digitalize, intelligent, paperless, self-operating ecosystems
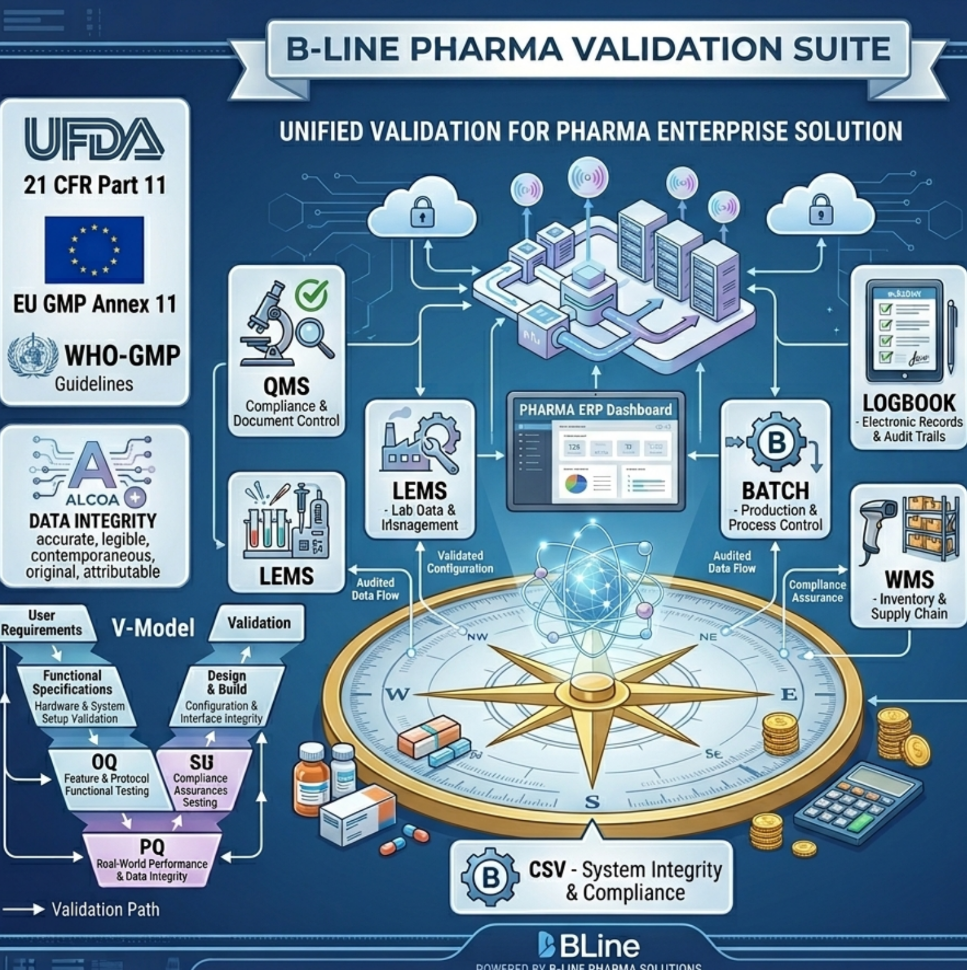
BLINE PHARMA SUITE: AI-Powered, GMP Compliance ready Pharmaceutical Enterprises Software & Hardware Solutions
Without Bline Pharma Suite:
– Batch records on paper lost, damaged, untrackable
– FDA inspector asks for batch history takes 2 days to compile
– Deviation happened CAPA done on Excel, no audit trail
– Equipment calibration missed nobody got the alert
– EU export rejected 21 CFR Part 11 non-compliance
– 5 different systems for QMS, LIMS, WMS – no integration
– Data integrity violation found in audit

With the Bline Pharma Suite:
– Every batch record digital, timestamped, ALCOA+ compliant
– Full batch history in 1 click — BMR report ready instantly
– Deviation auto-triggers CAPA with full electronic audit trail
– Calibration due alert 7 days in advance — zero misses
– EU & FDA regulated market exports — 100% compliant system
– One unified platform – QMS + LIMS + WMS + eBatch + IoT
– Always audit-ready – zero preparation time needed
Compliance with All Global Regulatory: USFDA – 21 CFR Part-11 | MHRA | TGA | ICH | PIC’S | SCHADULE-M | WHO-GMP Compliance | EU GMP Annex 11 | SFDA | MoHAP | MoH | EDA | NDA

BLINE FACTORY AUTOMATION SUITE 4.0
- Monitor every machine, process, and production line from a single dashboard. Get instant insights into production speed, quality metrics, equipment health
- Automatically manage production recipes, batch operations, and sequential processes. The system makes micro-adjustments to maintain optimal performance without human intervention.
- Connect any equipment old or new from any manufacturer. Supports EtherNet/IP, Modbus, OPC-UA, MQTT, and REST APIs, ensuring seamless communication across your entire facility.
- Never miss a critical issue. The system prioritizes alerts by severity and sends notifications to the right person
- Capture every data point from your production floor. Analyze trends, identify bottlenecks, and generate automated reports on OEE, downtime, quality
- Built-in validation support for pharmaceutical and regulated industries. Maintain complete audit trails, electronic signatures, and documentation for DQ, IQ, OQ, and PQ validation.
Transforming Manufacturing Through Intelligent Technology
we are your strategic partner in navigating the complexities of modern manufacturing. With deep domain expertise across automation, enterprise systems, artificial intelligence, and robotics, we architect integrated solutions that transform fragmented operations into synchronized, intelligent manufacturing ecosystems.
Manufacturing is no longer just about making products, it’s about making products smarter, faster, and with perfect traceability.
The factories that survive the next decade will be those that embrace:
✓ Connected ecosystems over isolated machines
✓ Predictive intelligence over reactive firefighting
✓ Flexible automation over rigid fixed lines
✓ Data-driven decisions over gut feeling
Years of Research
5+
Completed Projects
25+
Success Rate
98%
Countries
15+
Book a free 30-minute consultation. We’ll show you exactly where your gaps are, and how we closes them. 📞 Book Free Demo: Call Now