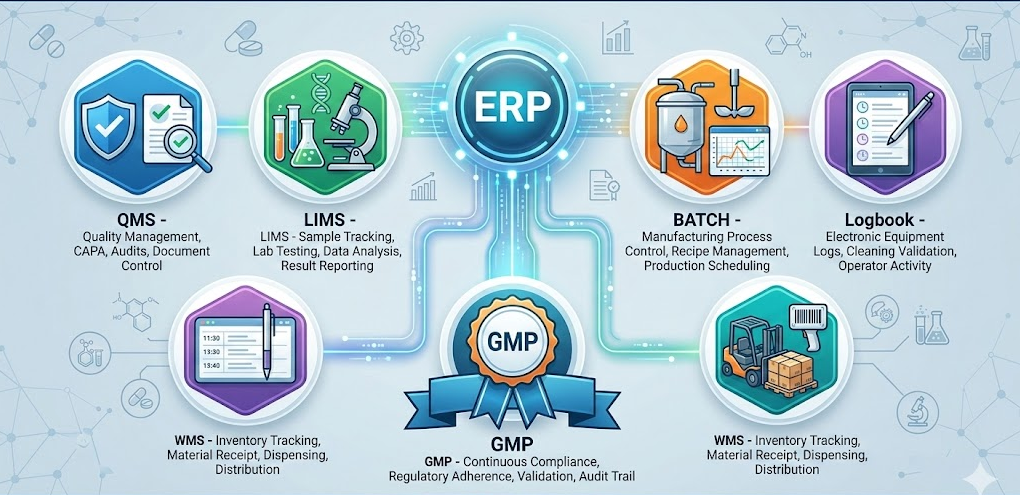
Why is Pharma ERP - a business necessity
Why Pharmaceutical Companies Can No Longer Afford to Operate Without an ERP System.
The pharmaceutical industry operates under some of the most demanding regulatory, quality, and operational standards in the world. Yet, surprisingly, a significant number of pharma manufacturers from growing mid-sized facilities to large, multi-site enterprises still working on disconnected system, or paper-based batch records, and many applications to manage their most critical processes. Size is no guarantee of integration.
Many large companies run separate systems for production, quality, lab, engineering, and inventory that do not communicate with each other, creating the same data gaps, compliance risks, and visibility blind spots as companies a fraction of their scale.
The Problem with Manual & Disconnected Systems
Data Duplication: The same batch data is written in logbooks, entered in paper, retyped in desktop software, and printed for QA review. Every duplicate entry is an opportunity for error. A single transposition mistake a misread quantity, an incorrect lot number can cascade into a deviation, a batch rejection, or worse, a recall. Multiply that risk across hundreds of batches per month, and the numbers become alarming.
Compliance Risk: When auditors from FDA, WHO-GMP, or EU GMP arrive, teams scramble to locate records. Missing pages, illegible handwriting, and mismatched data create 483 observations and warning letters. In a regulated environment, the inability to produce complete, traceable, and accurate documentation is not just operationally embarrassing it is a direct threat to your manufacturing license and market access.
Hidden Cost Leakage: Without real-time batch cost tracking, raw material wastage, yield losses, and rework costs remain invisible until month-end when it is too late to correct. By the time finance compiles the numbers, the damage is done. There is no opportunity to intervene, adjust processes, or hold suppliers accountable in the moment.
No Real-Time Visibility: Plant managers and QA heads have no live view of production status, batch progress, or quality deviations. Decision-making is always reactive, never proactive. Issues that could have been caught at the source are instead discovered downstream during QA review, during packaging, or worse, after dispatch.
Delayed Batch Release: Manual review of paper BMRs (Batch Manufacturing Records) takes days to weeks. Batch release is delayed, impacting customer deliveries and market commitments. In an industry where on-time supply can determine whether a hospital shelf is stocked or empty, that delay carries both financial and human consequences.
Industry Research: A pharmaceutical company loses an average of 25-40% of operational efficiency due to manual documentation and disconnected systems.
