BLine OmniMind AI Suite
The AI Intelligence Layer for Your Enterprise
BLine OmniMind AI is a next-generation, fully controlled artificial intelligence platform purpose-built for regulated, data-sensitive industries including pharmaceutical research, drug discovery, industrial automation, healthcare, and specialized research environments.
All infrastructure, model weights, data pipelines, security architecture, and operational systems are proprietary assets of Bline Solution Pvt. Ltd., delivered under a strictly governed service and licensing model.
Hybrid Intelligence Capability
Where required, OmniMind supports secure, policy-controlled API integrations with external AI systems such as Claude. These integrations are architected to ensure:
No unauthorized data exposure
Full auditability and traceability
Strict compliance with enterprise governance policies
Optional air-gapped or proxy-mediated communication layers
This enables enterprises to leverage external intelligence models for advanced reasoning or enrichment—without compromising data sovereignty, security, or regulatory compliance.
What BLine OmniMind AI Can Do?
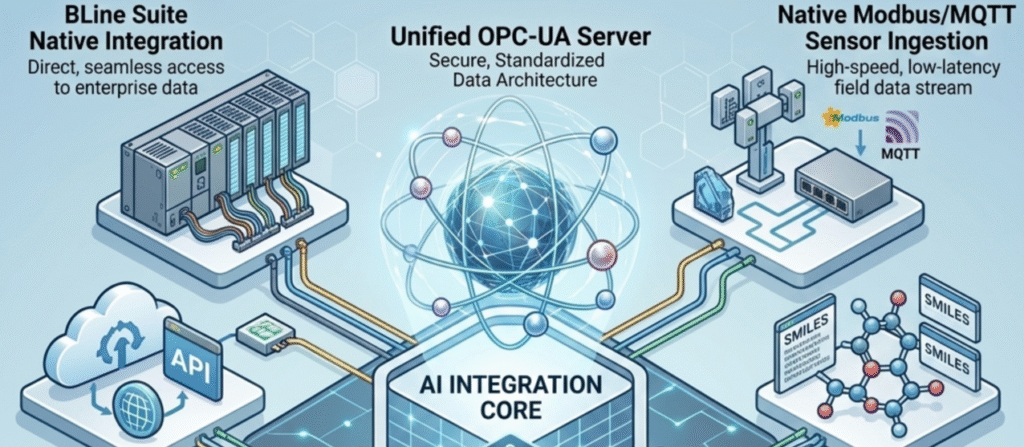
BLine Suite Integration: Functions as the AI intelligence layer across BLine Pharma Suite and BLine Automation Suite—automating workflows, batch control, LIMS queries, plant automation, and security integration with full RBAC compliance.
Drug Discovery & Safety:
Protein structure analysis (AlphaFold-style), molecular property prediction from SMILES, drug-drug interaction forecasting, adverse event prediction, and biomedical literature synthesis—all within controlled environment.
Industrial Automation:
Real-time quality inspection, defect detection, PLC ladder logic generation, process optimization, predictive maintenance with MQTT/Modbus sensor feeds, and equipment health scoring.
Scientific Applications:
New material design, reaction prediction, spectroscopy analysis, automated interpretation integrated with BLine LIMS for result logging and CoA generation.
Regulatory Compliance:
Automated generation of CTD submission modules, regulatory dossiers, audit reports, CAPA documentation, and GMP compliance records—formatted for FDA, EMA, and ICH standards.
Adaptive Learning Platform:
AI-personalized training grounded in organization’s actual SOPs, regulatory frameworks, and batch processes—with assessment generation, competency tracking, and multilingual delivery.
